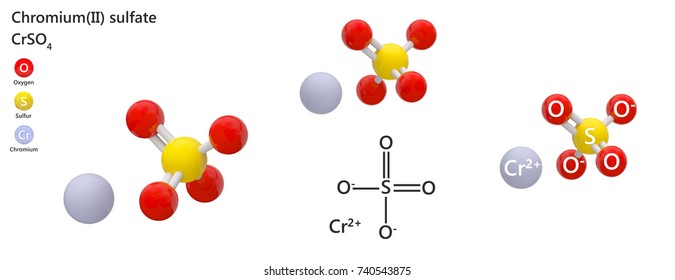
When we write the two together we get Cr 2 (SO 4 ) 3. So, to arrive at the formula of Chromium III Sulfate, we will interchange the charges of ions Cr +3and SO 4 -2 to get Cr 2 and (SO 4 ) 3.Cr is without parenthesis which indicates it is not a polyatomic ion. The sulfur and oxygen atoms are inside brackets indicating a covalent bond between them. Chromium(III) sulfate is a compound of chromium and sulfate in which the ratio of chromium (in the +3 oxidation state) to sulfate is 2:3 It contains a chromium(. 
Cr is placed outside the parentheses as it is not a polyatomic ion. Chromium III Sulfate Cr2(SO4)3 is an ionic compound where Cr is the metal with Cr+3 cation and sulfate is the SO4-2 anion. Potassium chromium sulfate, KCr (SO 4) 2 12H 2 O, or chromium alum (1), forms violett octaeders and is highly soluble in water. The roman numeral tells us that the positive charge in this case is +3, so. So, polyatomic ion SO 4 -2 will be placed inside parenthesis. A Because chromium is a transition metal, it can form cations with different charges. The sulfur and oxygen atoms share a covalent bond.Names of Some Ionic Compounds NaCl, sodium chloride Na2O, sodium oxide KBr. So, the Charge carried by the chromium cation Cr +3 will be written as the subscript of the sulfate anion SO 4 -2 and the Charge carried by the sulfate anion SO4-2 will be written as subscript for the chromium cation Cr +3. We will limit our attention here to inorganic compounds, compounds that are.Chromium III has a valence of 3 and Sulfate has a valence of 2, we will need 2 chromium and 3 sulfate ions to balance 6 valence ties.It is an ionic compound where Chromium (Cr) is the metal with Cr+3 cation and sulfate is the SO4-2 anion. The formula of Chromium III Sulfate is Cr 2(SO 4) 3.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
